Interactive Dashboards Part II - Activating Smart Dashboards
- The Smart Dashboards are a paid add-on. The price varies by subscription plan (due to the amount of data/bandwidth needed to load the larger amounts of data at higher record counts):
- Easy Launch - $10/per user/month
- Easy Start - $10/per user/month
- Easy Grow - $10/per user/month
- Easy Go - $20/per user/month
- Easy Pro - $30/per user/month
- Every user can try the Smart Dashboards for free for 15 calendar days. All users can self-activate the 15-day free trial. However, only system admins can activate Smart Dashboards permanently (upgrade to paid use) for each user.
- Users must have permission to View Donations to see the two Fundraising Dashboards, View Events to see the Events Dashboard, and View Memberships to see the Membership dashboard. All user permissions are set in the My Accounts: User Roles area of your system. Users can be given permission to 1,2, or all 3 of the Smart Dashboards (e.g. just membership or just events dashboard(s) or to both membership and events dashboards, etc.)
- To activate the 15-day free trial, select Dashboard from the Main Menu. This will take you to the Basic (regular) Dashboard. In the upper right, you will see a toggle for Basic versus Smart Dashboard. When users first land on the Dashboard, a pop up bubble will appear encouraging users to try the Smart Dashboard. Click Try to activate the 15-day free trial.
- If user clicks "Later", the bubble disappears. Users will not see this bubble again. However, users can still try the Smart Dashboards simply by toggling the slider in the upper right to Smart Dashboard.
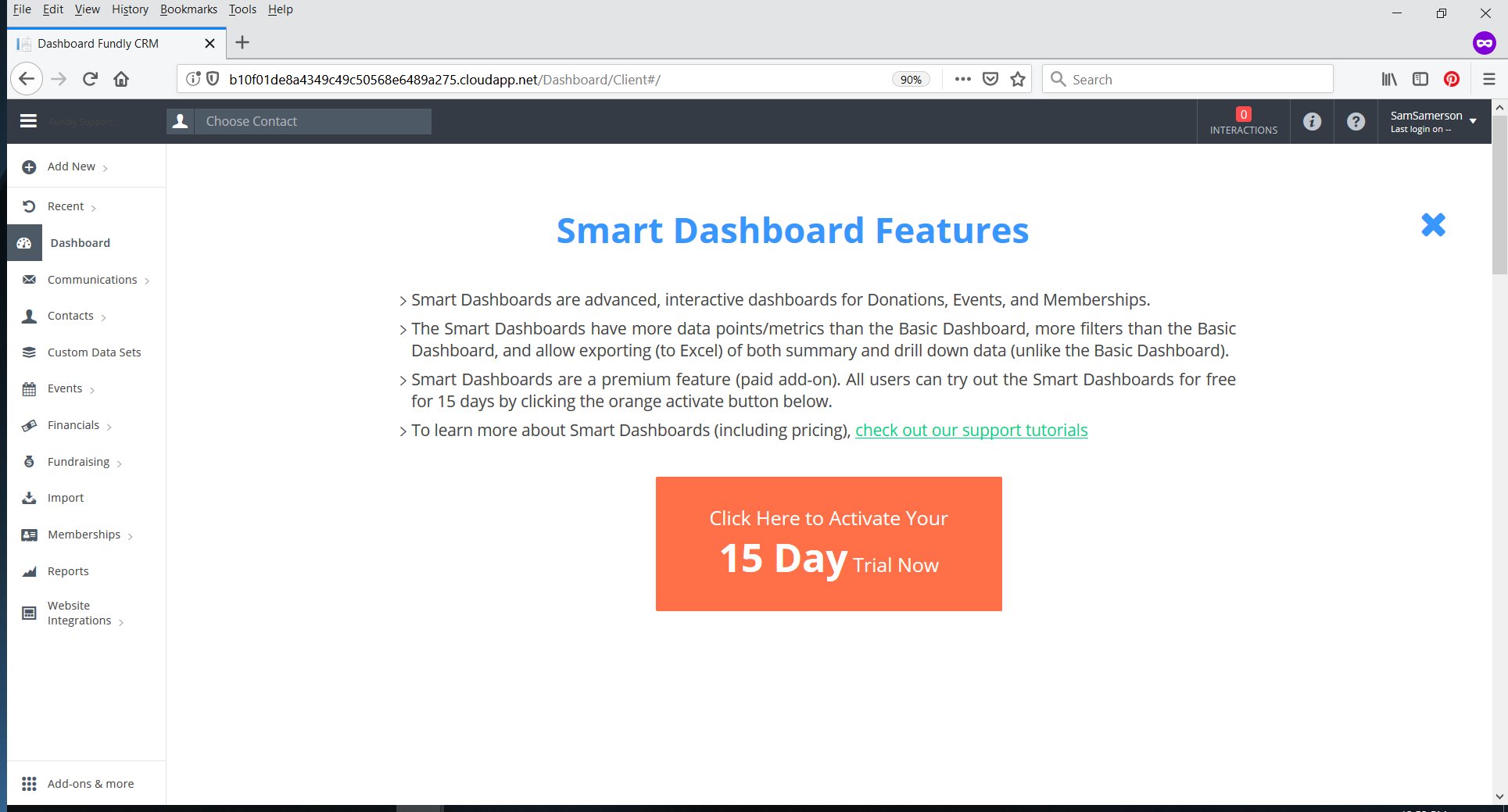
- On the Activate 15-Day Trial page, click the orange Start 15-Day Trial box. If you are a non-admin user, you will only see that box.
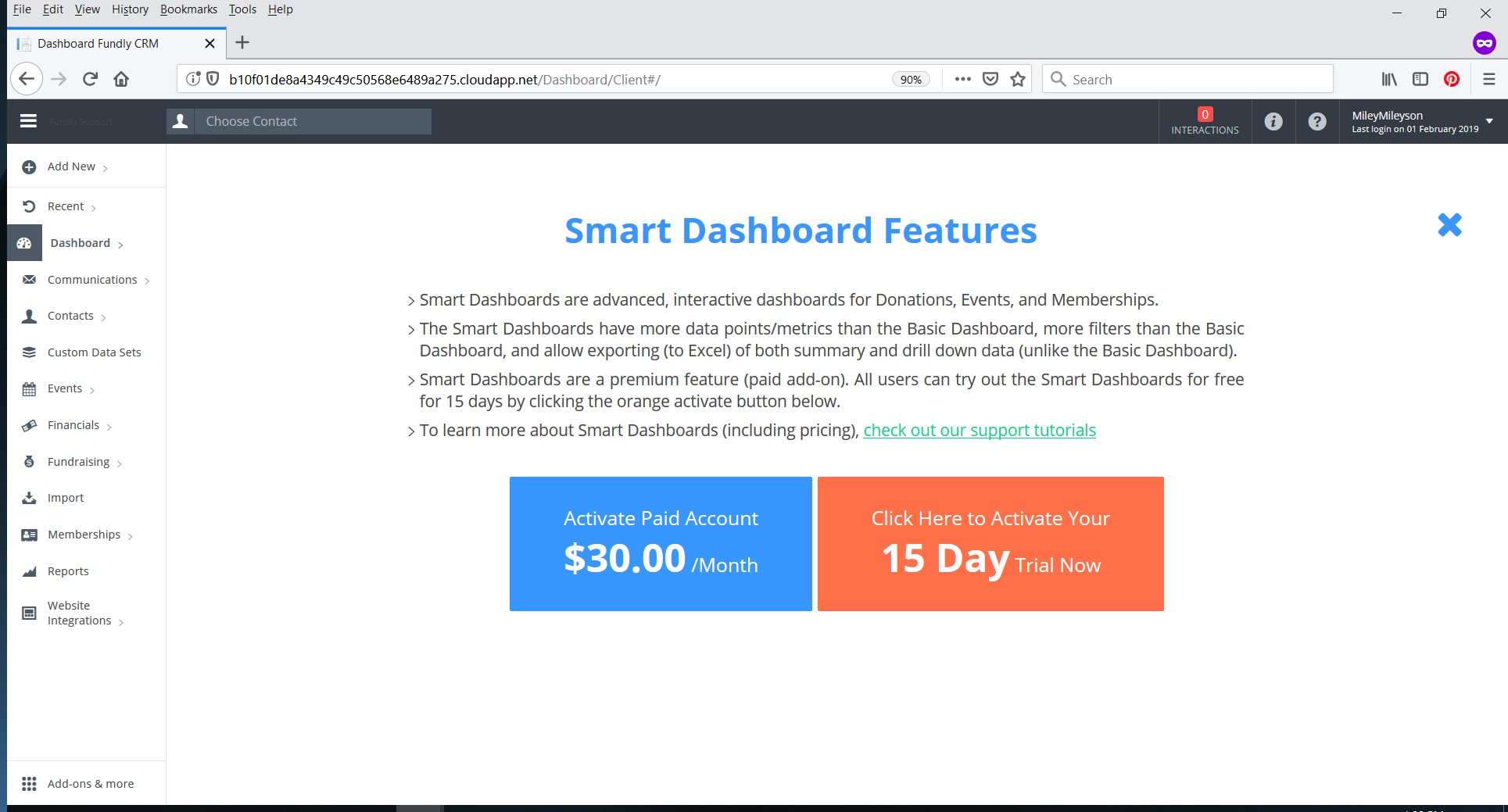
- If you are a system administrator, you will see that box plus a blue box to activate a paid account for yourself (bypass the free trial and go straight to a paid account).
- On the next page, click Confirm if you want to activate the 15-Day free trial or Cancel if you want to cancel and activate it later. Once activated, the trial is only good for fifteen (15) calendar days. There is no way for either your system admin or CRM technical team to pause or extend the free trial, so be sure you will have adequate time to try the dashboard before activating the free trial.
- You will see a confirmation screen alerting you that your free trial has been activated. In addition, your system administrator will receive an email, alerting them that you have activated the free trial.
- Click the Continue button to proceed to the Smart Dashboard.
- You will be taken directly to the Fundraising Smart Dashboard. Now and for the next fifteen days, you will see a new sub-menu on Main Menu: Dashboard that will allow you to access the Fundraising, Event, and Membership Smart Dashboards if you have the rights/permissions to those dashboards (Note: Non-Admin users will not see the Manage Users section of the Dashboard sub-menu).
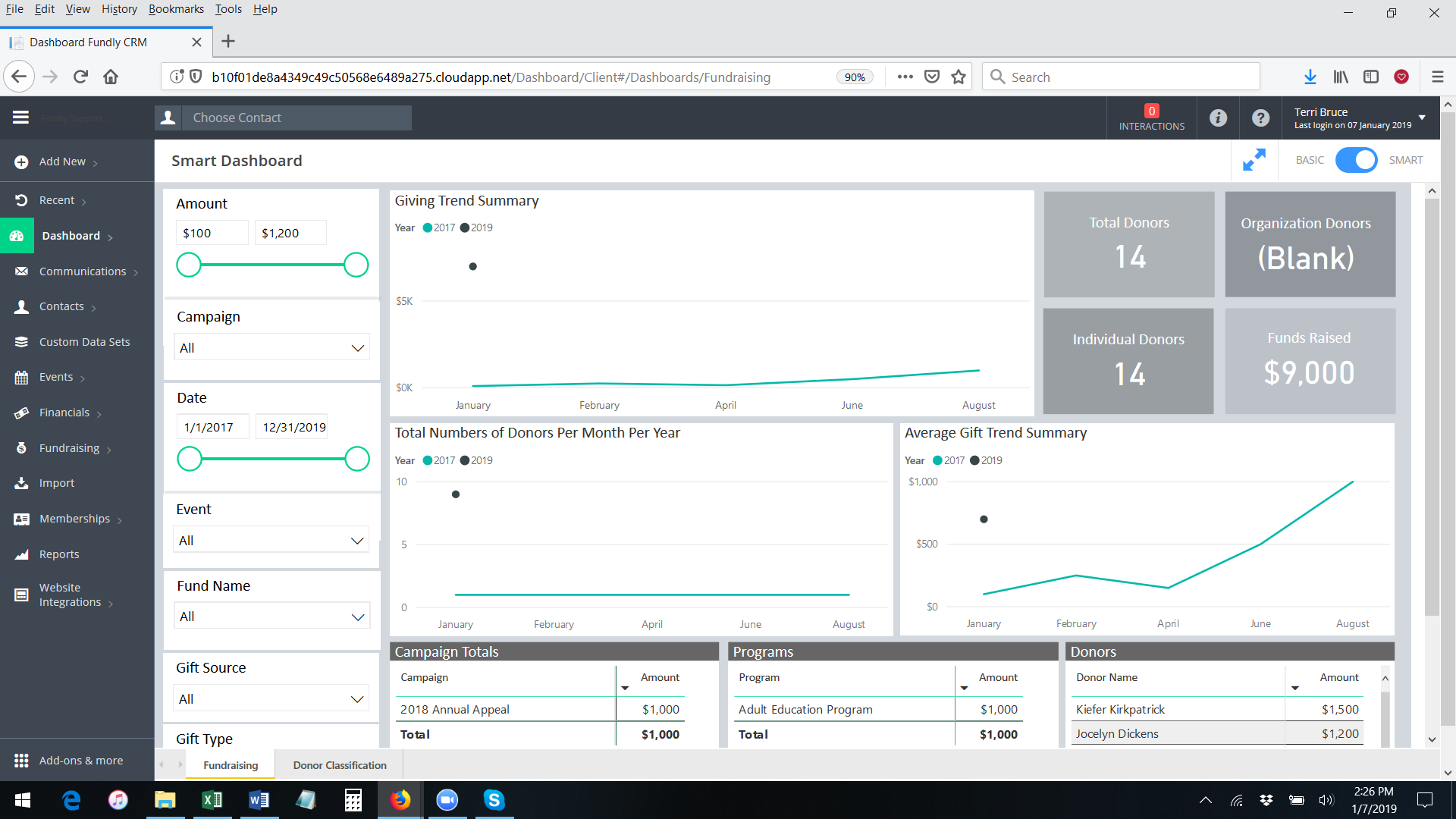
- In addition, you will be able to toggle back and forth between the Basic Dashboard and the Fundraising Smart Dashboard using the toggle in the upper right hand corner of any Dashboard screen.

- You will also see how many days you still have left in your free trial.
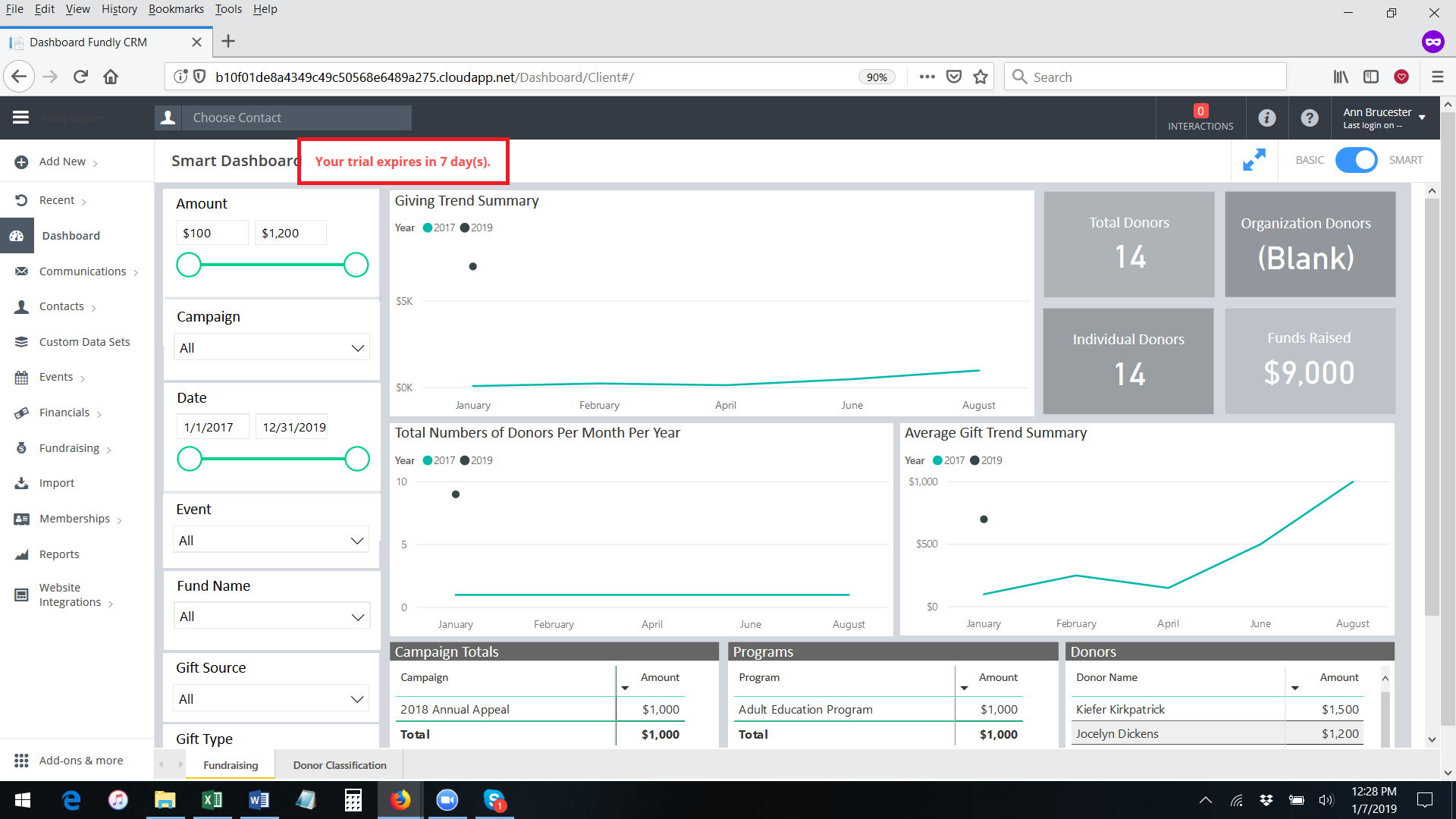
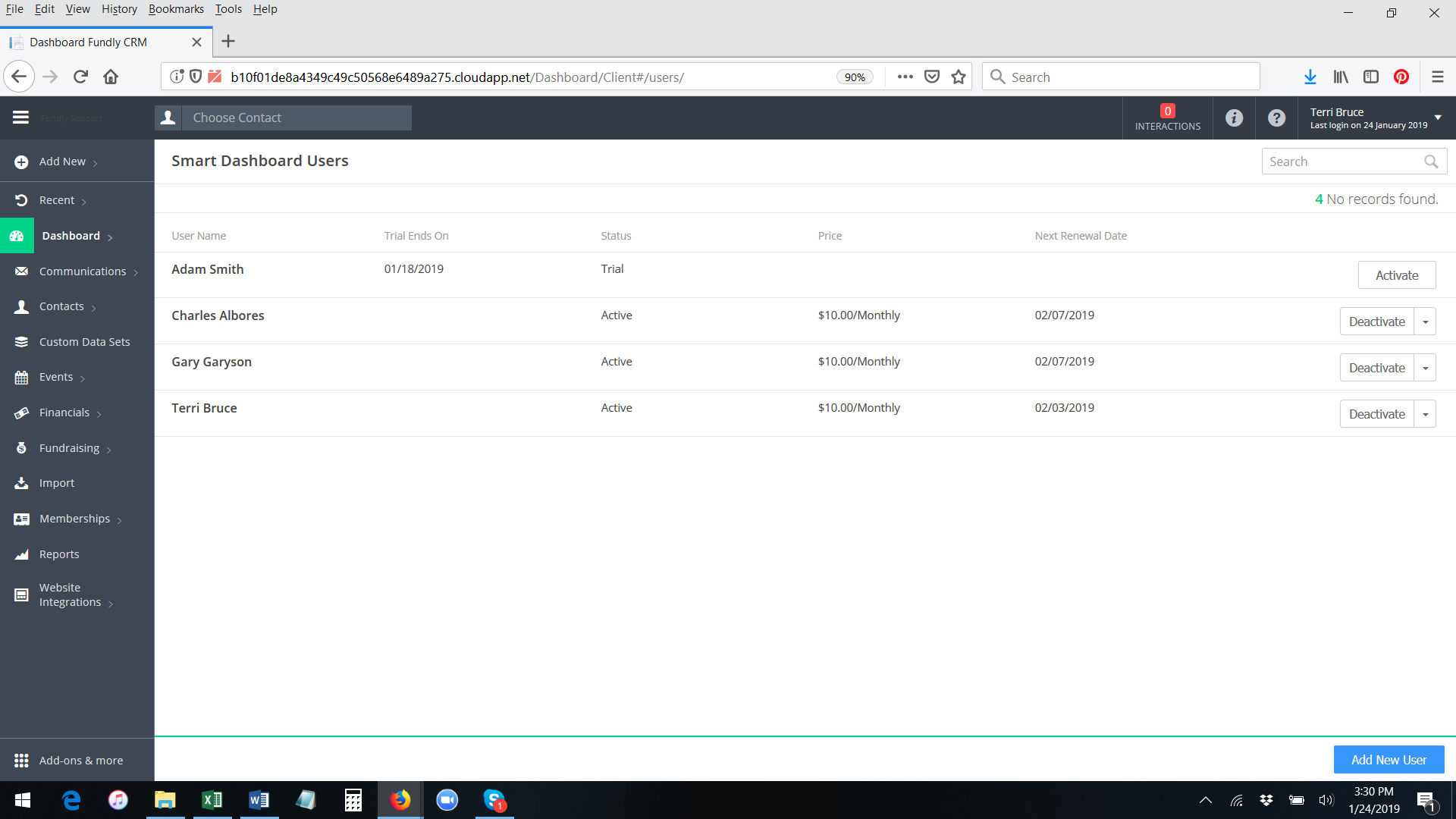
- To permanently activate the interactive dashboards for any user (this can be done by system admins ONLY), go to Main Menu: Dashboard and select Manage Users (only system admins will have this option).

- This will show a list of users who have activated smart dashboards and the status of smart dashboards for each of them (active trial, expired trial, active (paid), deactivated (paid)).
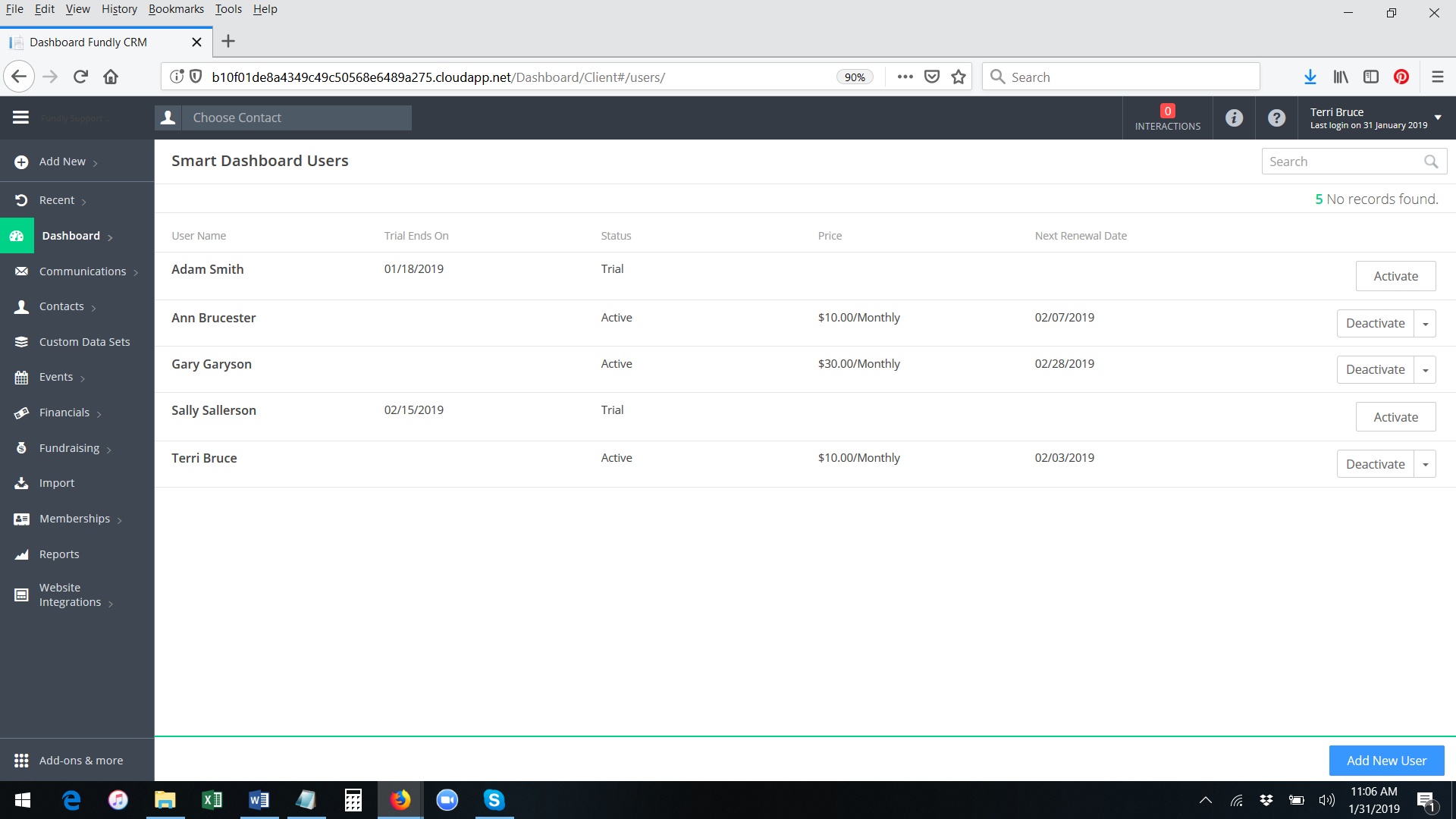
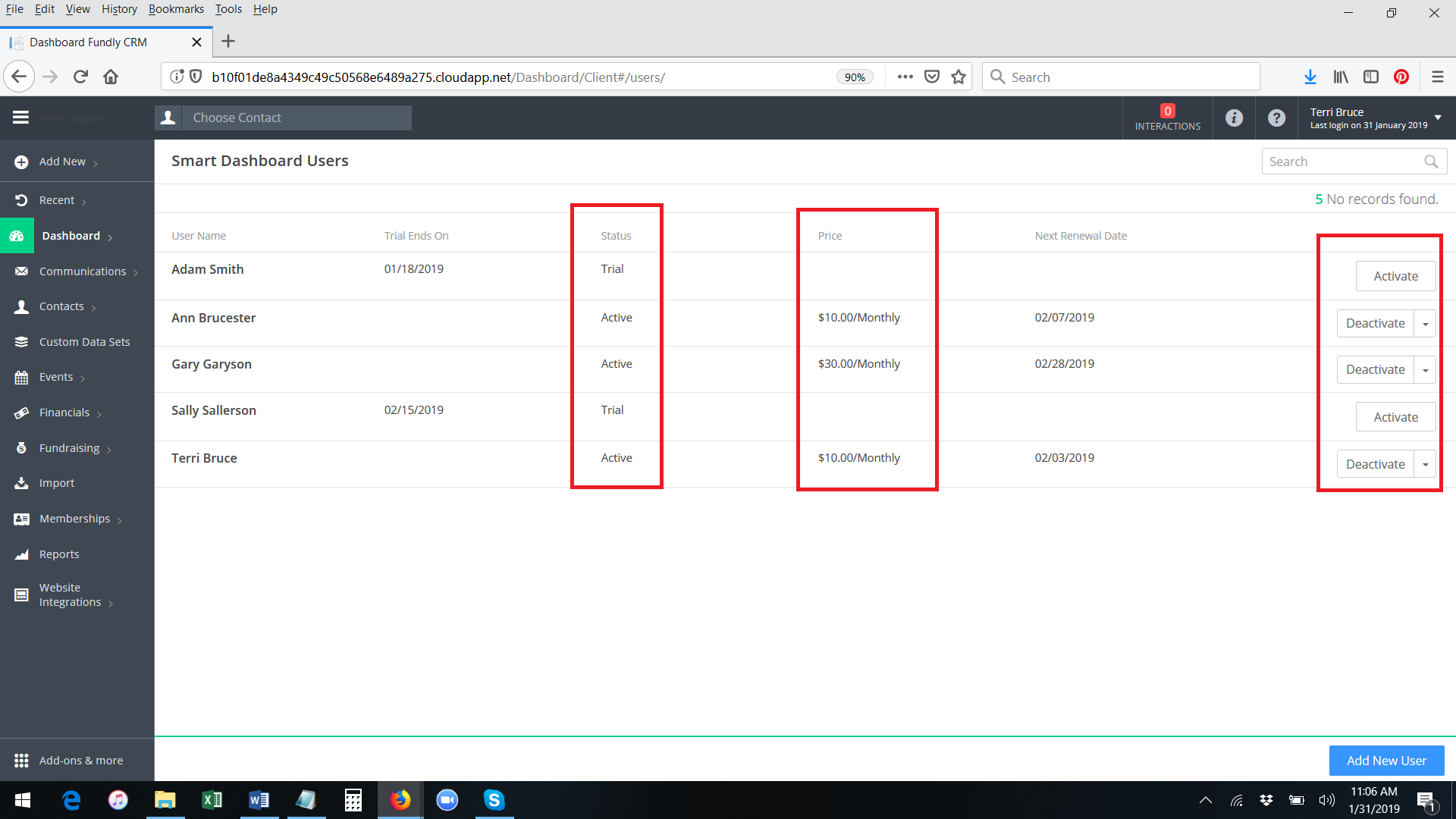
- To activate a paid subscription/permanent access to the Smart Dashboard for any user who has already activated the free trial, click the Activate button to the right of the user's name.
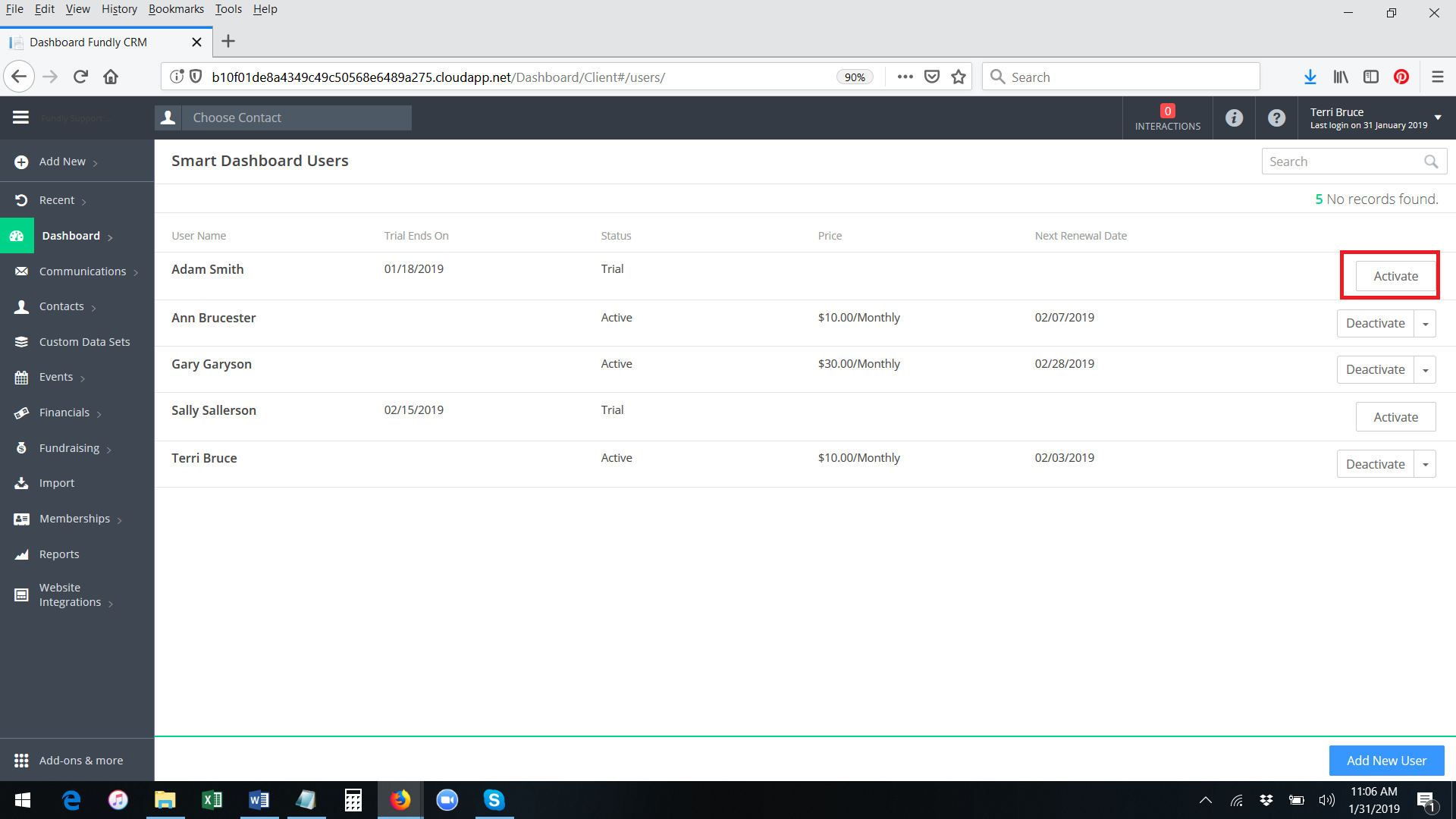
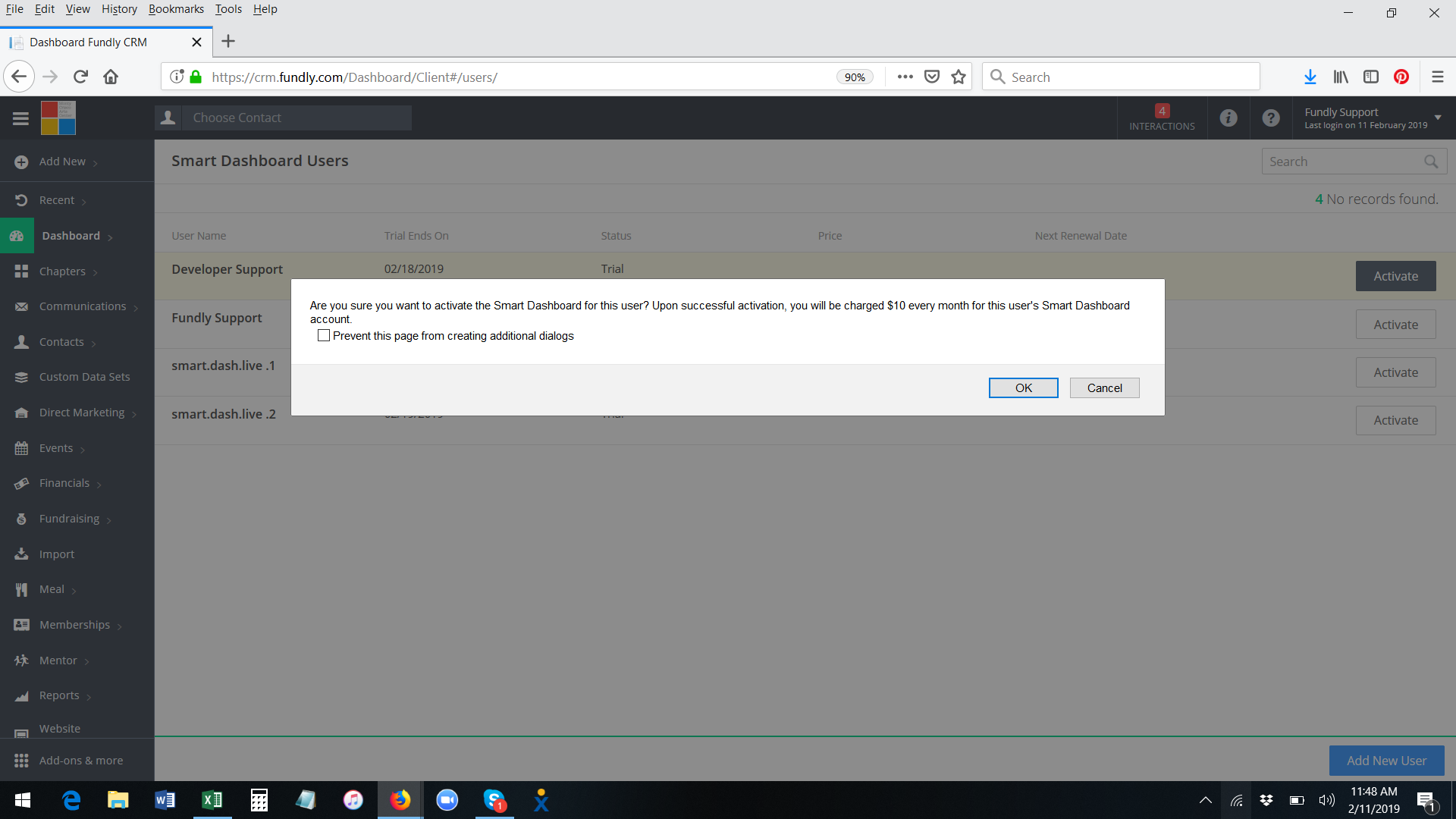
- A pop up message will appear, asking you to confirm the activation and displaying the amount you will be charged for this user (Smart Dashboard price is per user per month). The credit card on file will be charged immediately upon activation for the first month and then every month thereafter on the same day (anniversary date). Click OK to confirm.
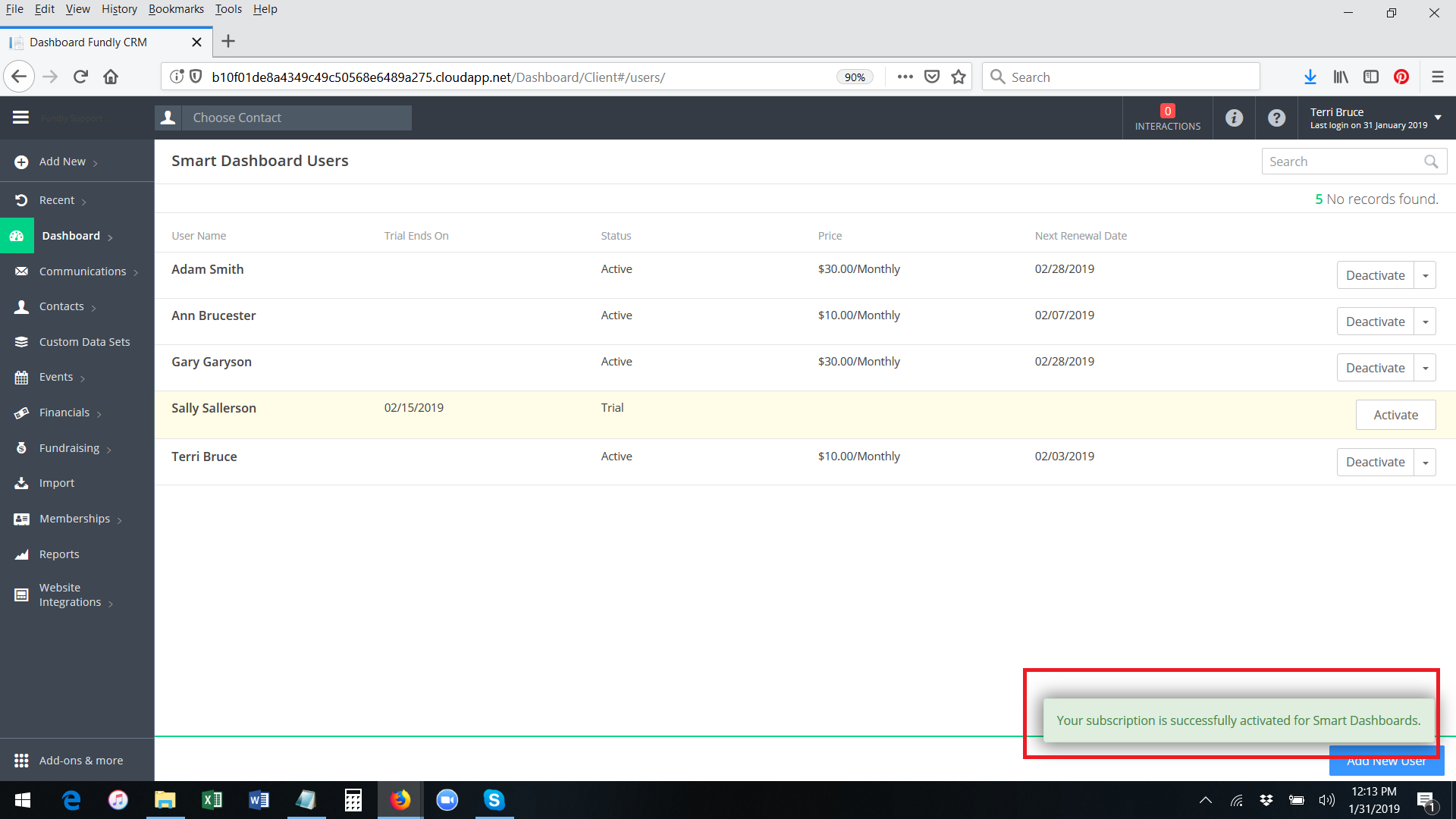
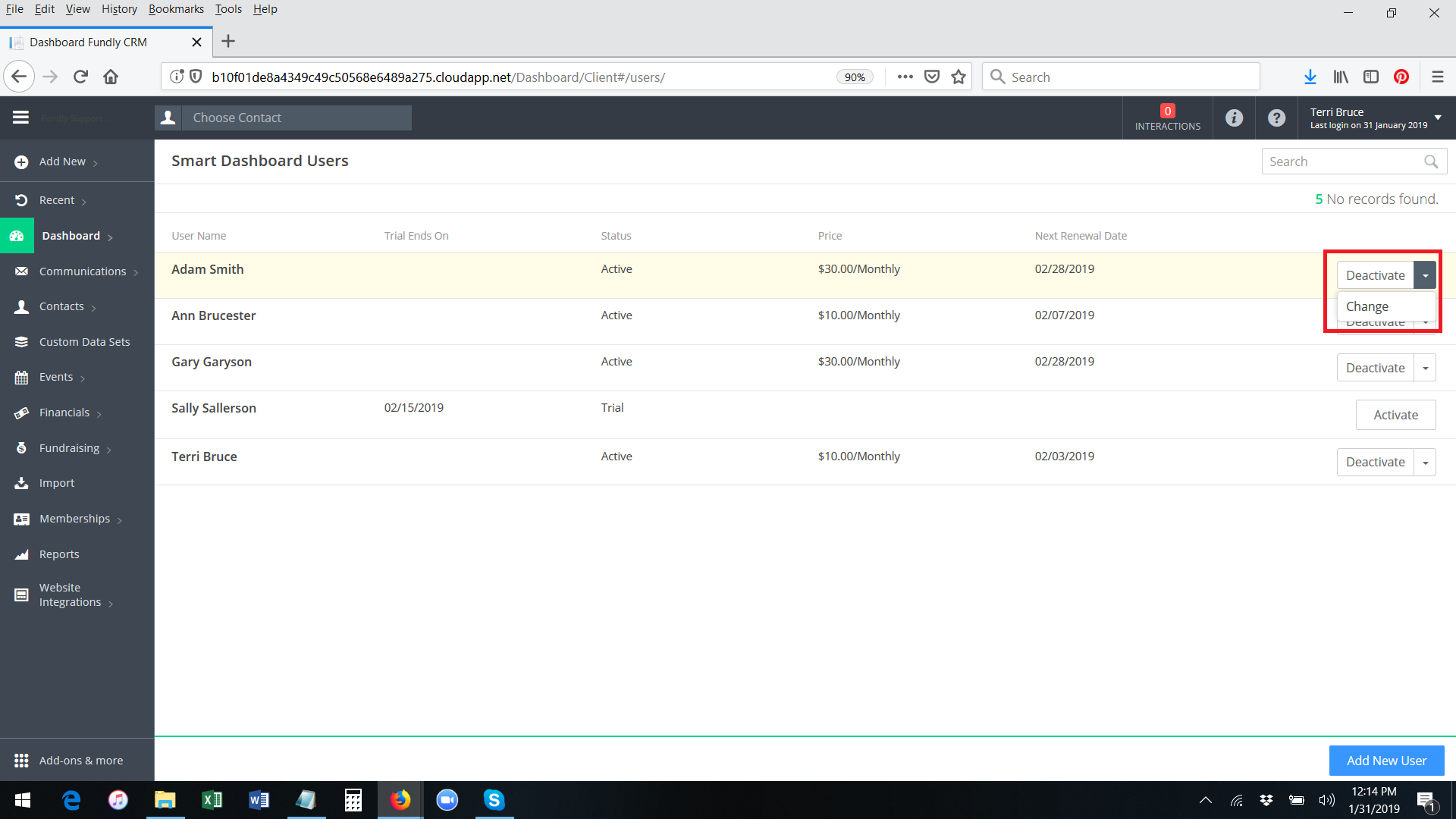
- The system will permanently activate the smart dashboard for the user. You will receive a green validation message in the lower right hand corner of the screen. The button to the right of the user's name will change to show Deactivate and Change as the options.
- If you wish to activate the Smart Dashboard subscription for someone who has not yet activated the free trial (if you want to bypass the free trial), click the blue Add New User button in the lower right hand corner of the list of the Smart Dashboard Users.
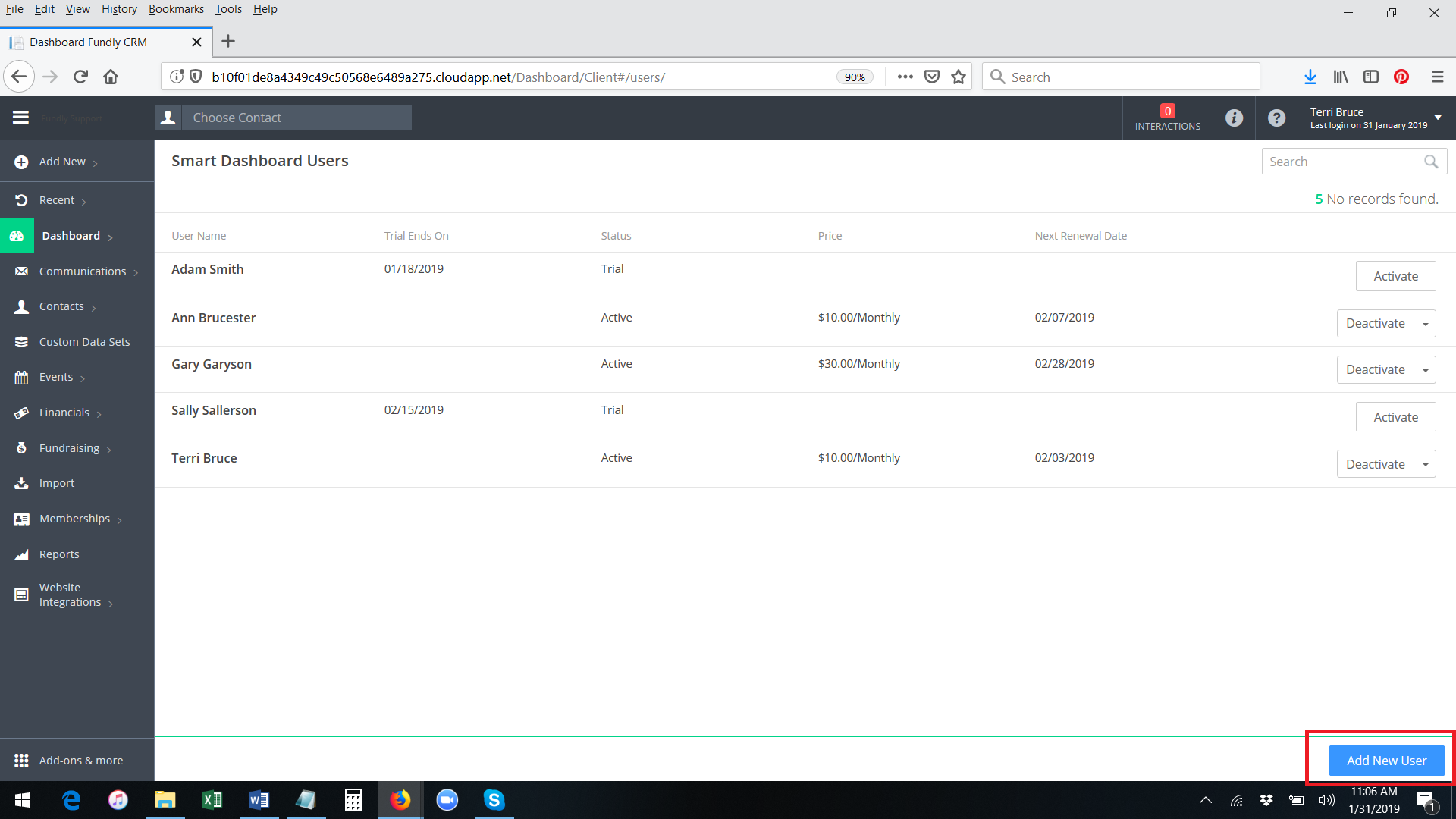
- Start typing the User Name of the user you wish to activate Smart Dashboards for. The system will display a list of users that match what you type. Please Note: If the user already has a paid Smart Dashboard account activated, their name will not appear in the list of matching results. Only those users who do not already have a paid Smart Dashboard account will appear.

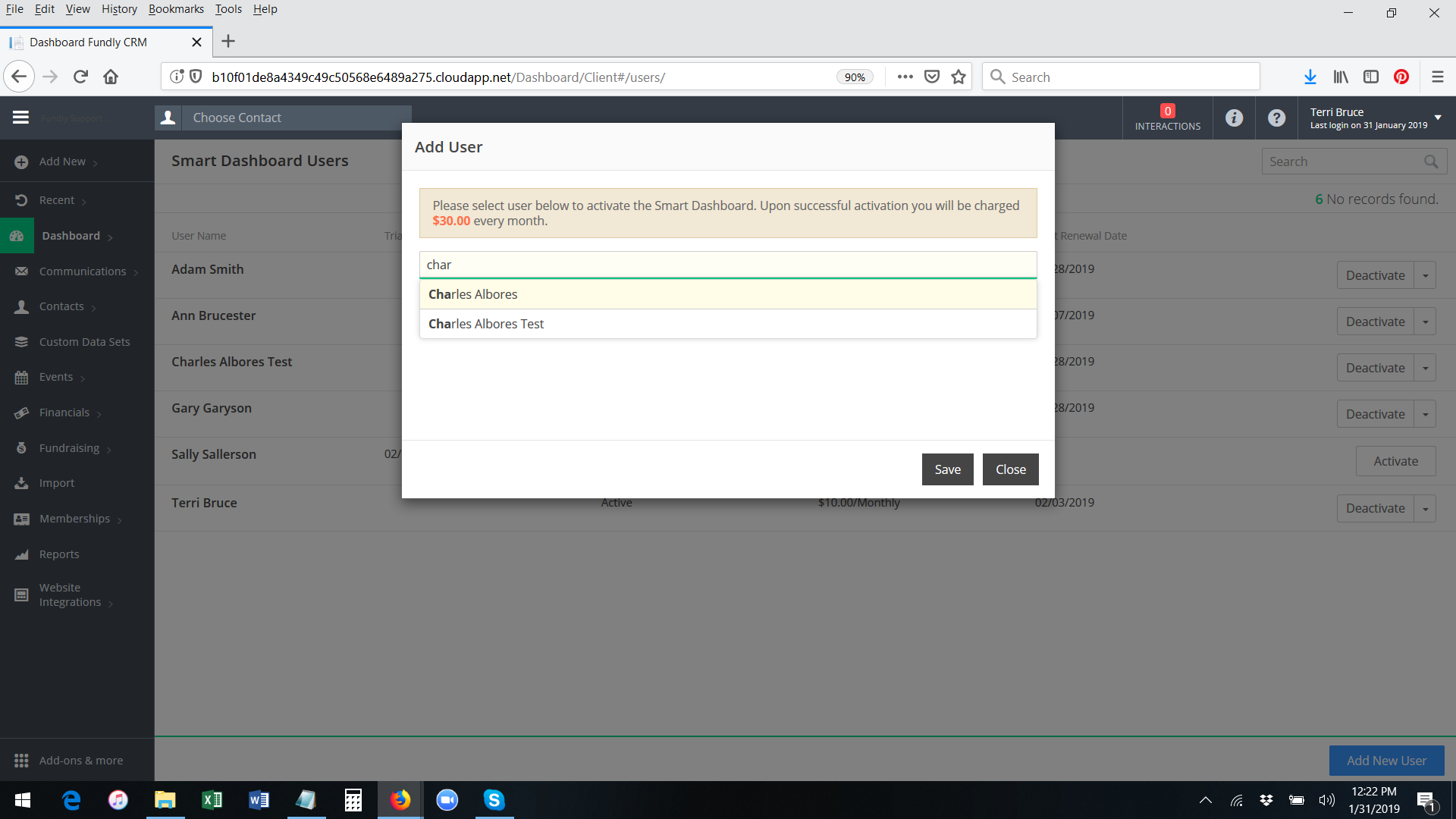
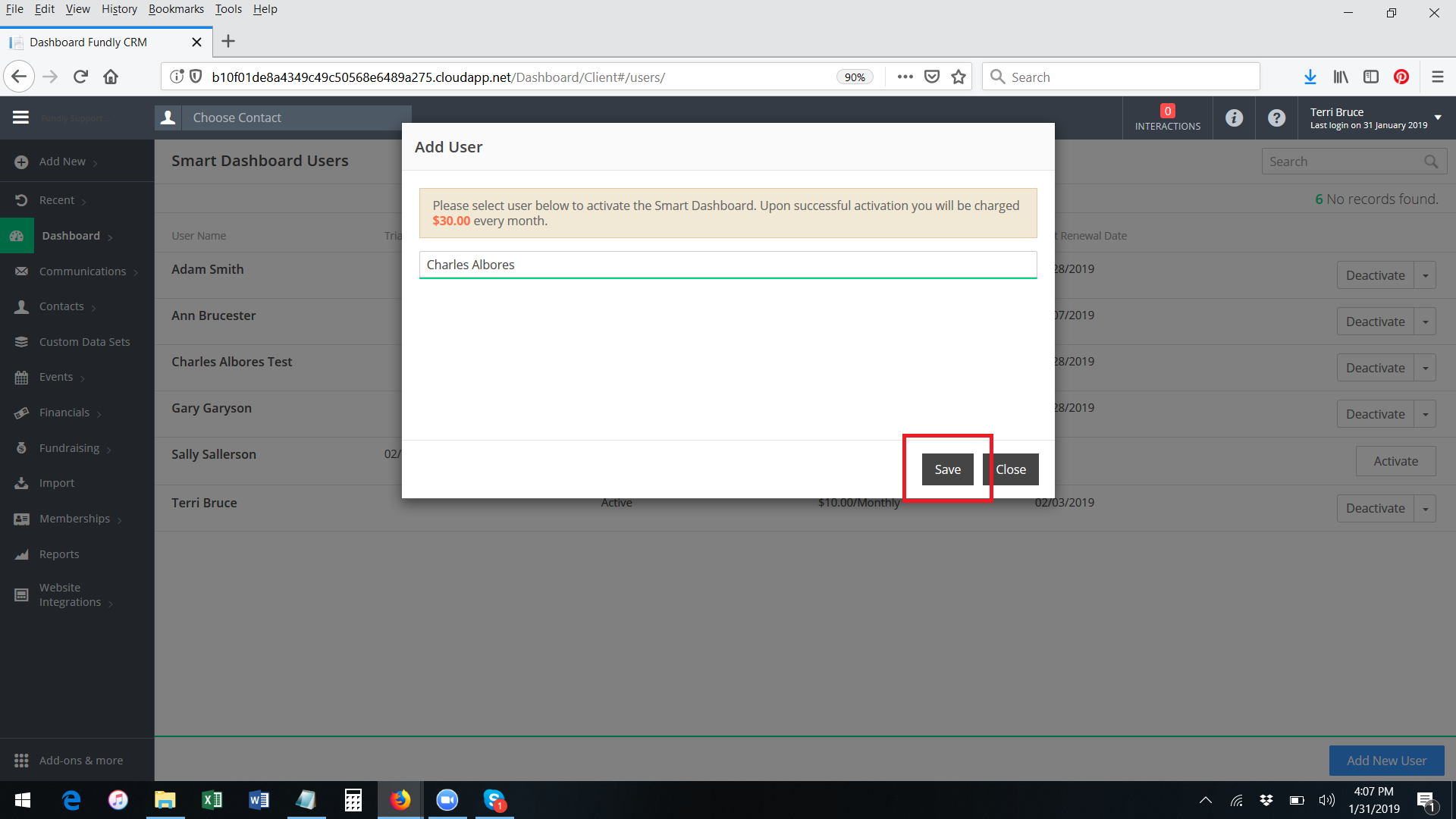
- Select the correct user from the list and click Save.
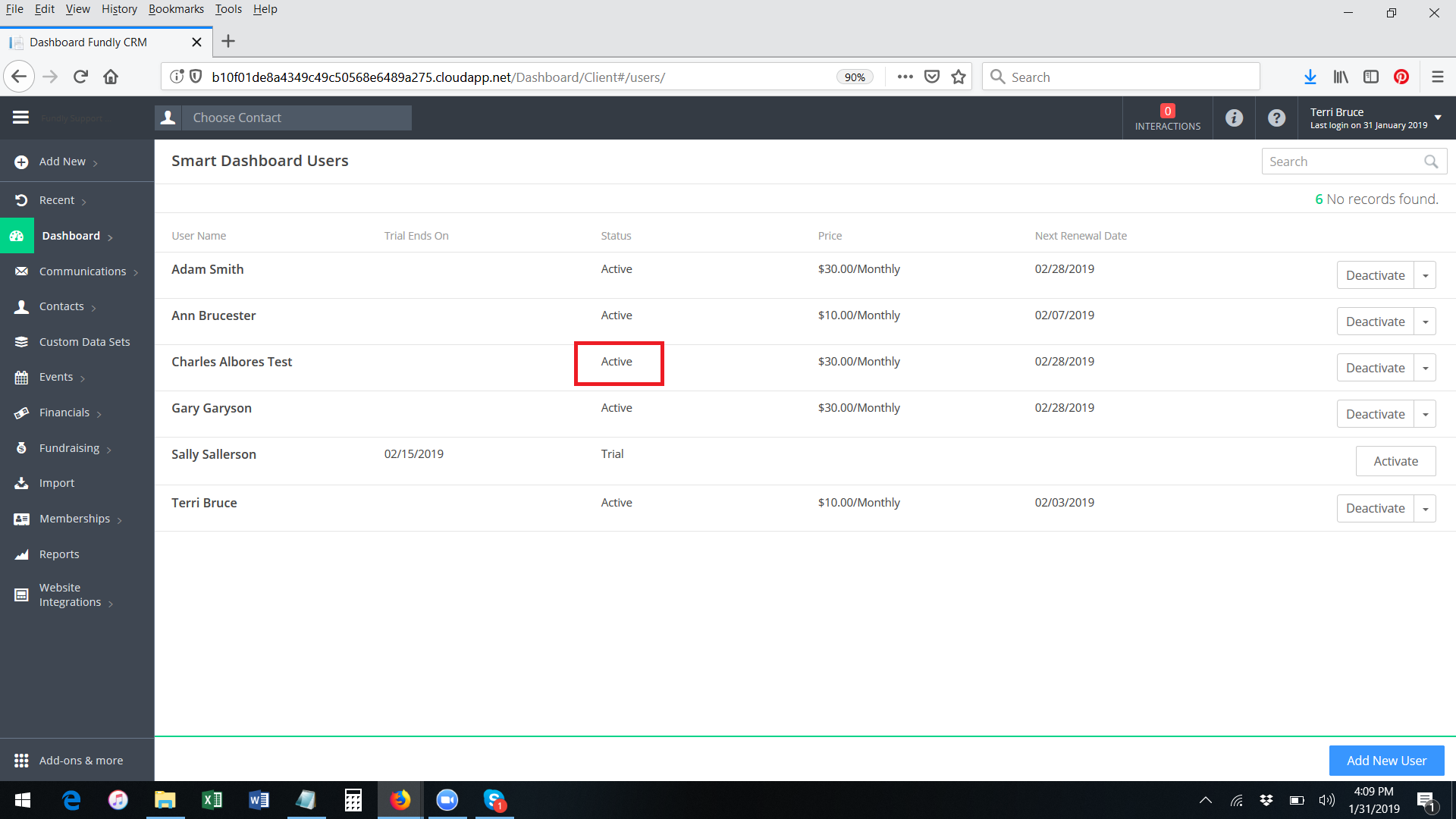
- The User will be added to the list of Smart Dashboard Users and will show a status of "Active" - this process bypasses the free trial step and activates a paid subscription for the user.
- If you want to swap one active Smart Dashboard user for a different one (that is, if Ann has an active/paid subscription to the Smart Dashboards and no longer needs it and you want, instead, to activate a paid subscription for James) you can use the Change function. (that is, rather than going through the process of deactivating Ann's Smart Dashboard account and then activating an account for James, you can simply replace James for Ann by using the Change function; this will keep the same billing cycle that Ann was on and will prevent you from having to pay twice in the same month - once for the user you are deactivating and once for the user you are activating). On Main Menu: Dashboard: Manage Users, find the user in the list of active users that you want to replace/deactivate. Click the drop down to the right of the user's name and select Change.
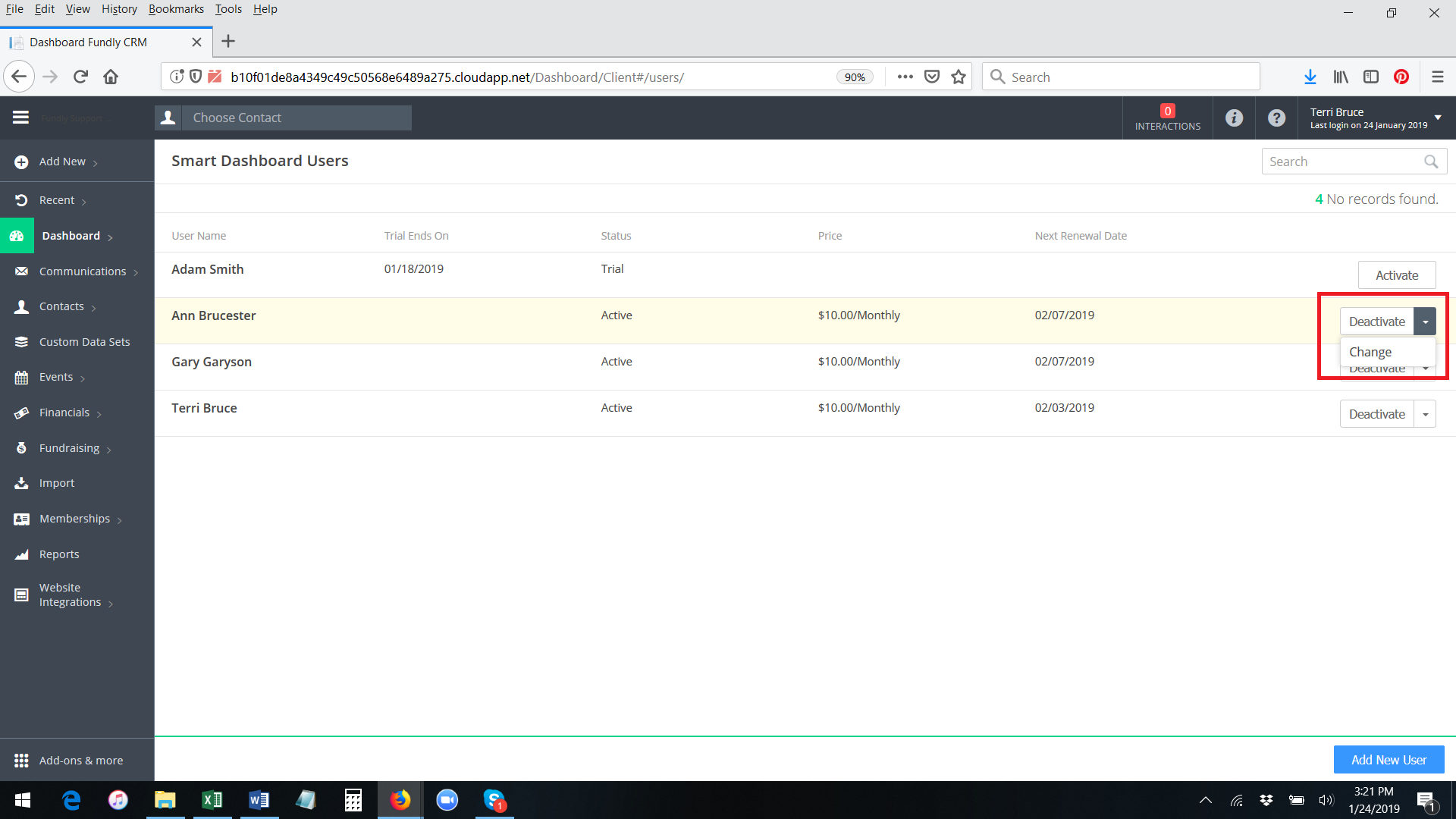
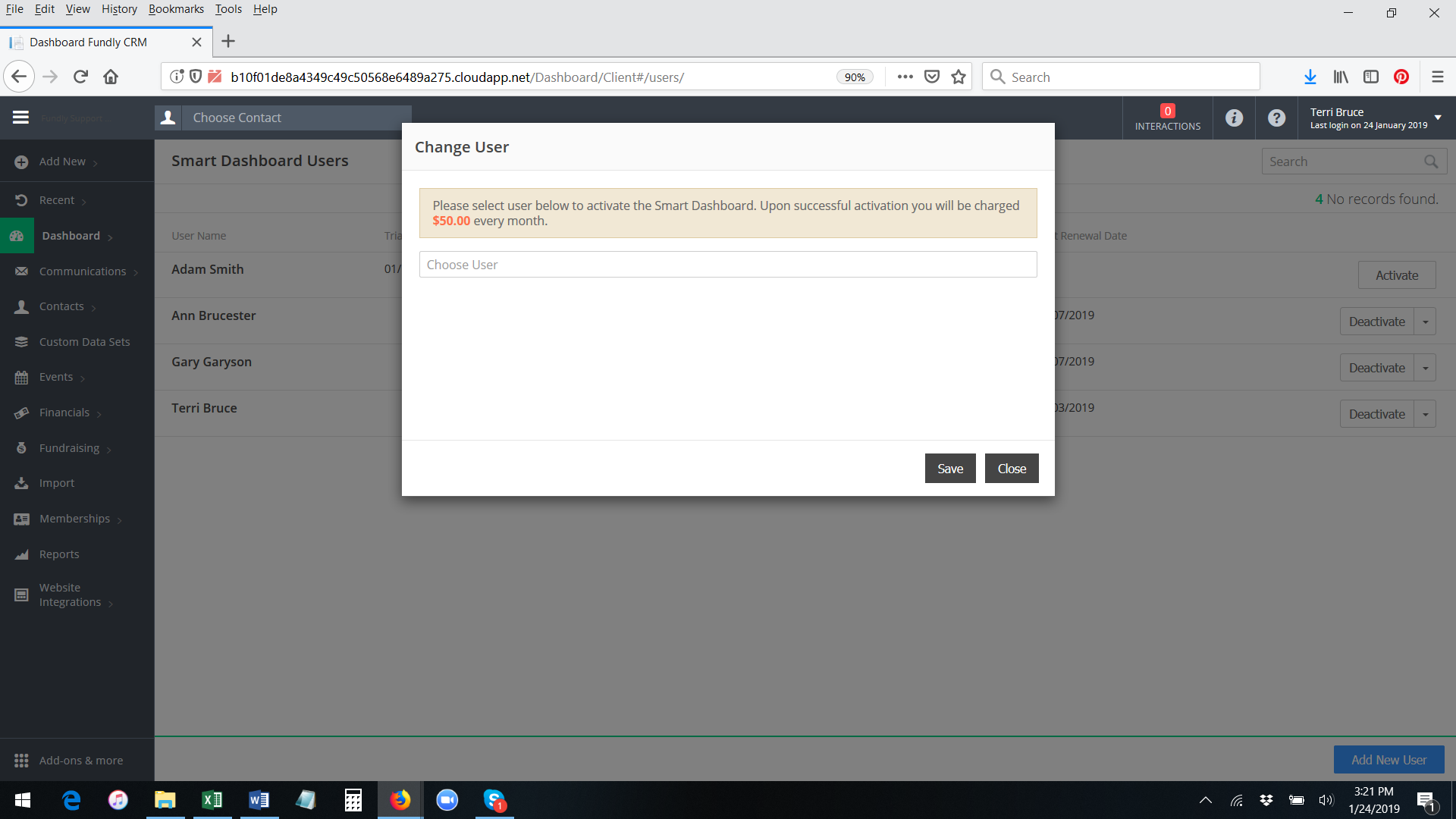
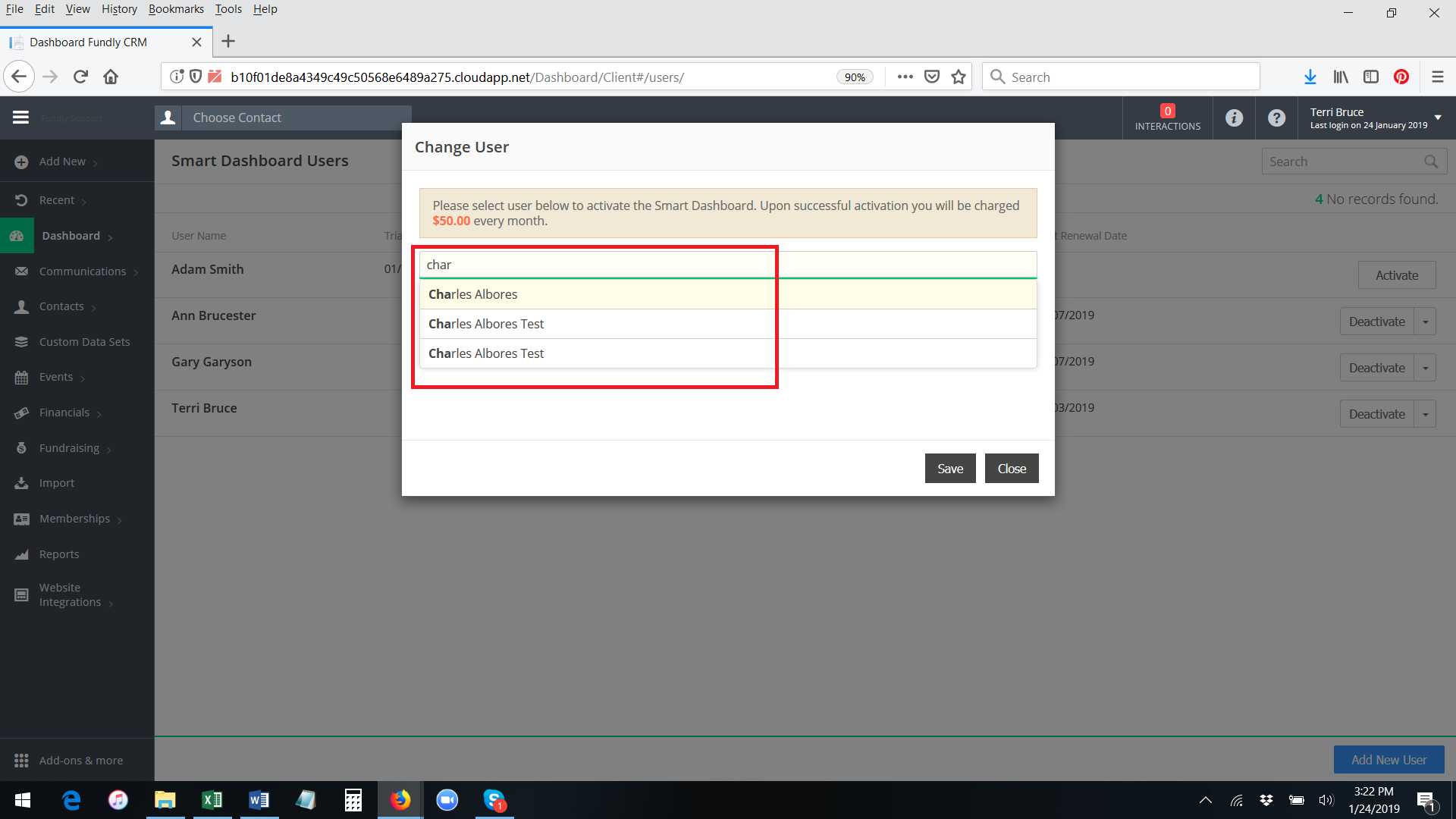
- Start typing the user name of a user already configured in your system and then select off the list of matching names. Then click on change to Save.
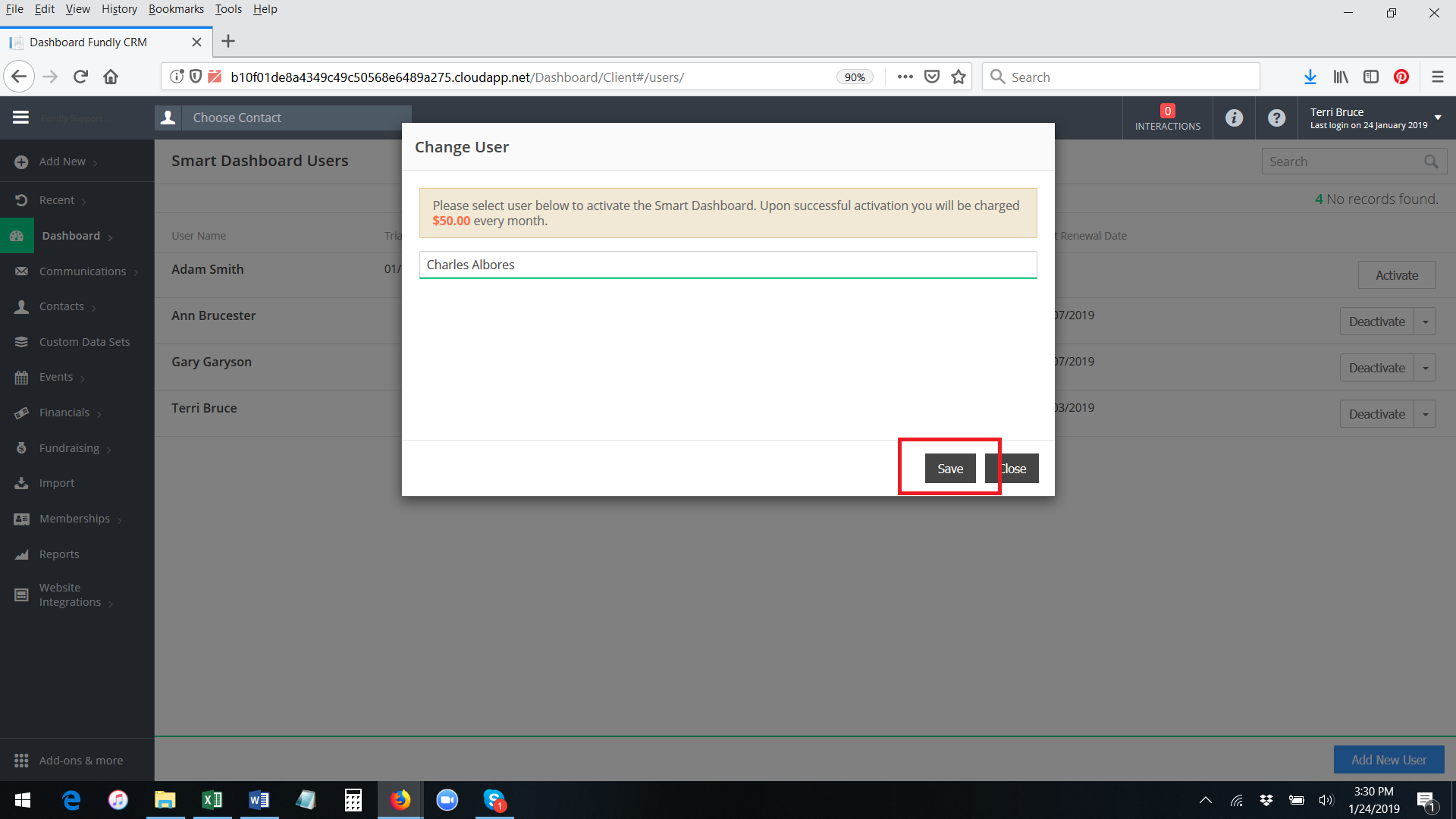
- Now the new user will have replaced the old user in the list of Smart Dashboard users.

Comments
0 comments
Article is closed for comments.